Introduction
Lumen are cavities that extend into the body.
There are four main fluid compartments:
- Intracellular fluid inside of cells
- Interstitial fluid outside of cells and blood vessels
- Plasma inside of blood vessels
- Glycocalyx
Homeostasis
Homeostasis is the ability to keep the internal environment within a tolerable range.
Feedback systems maintain homeostasis. In a positive feedback system, stimuli is received by the receptor, which sends the information to the integrating center, which sends instructions to the effector, which carries out the response. For example, a drop in blood pressure causes the heart to speed up and the kidneys to retain fluid.
Cell Physiology
Mitochondria converts sugar, fats, proteins, and O2 into ATP and CO2 through cellular respiration.
The cell membrane is a protein-studded phospholipid bilayer. The head of the phospholipid is polar (hydrophilic) and the fatty tails are nonpolar (hydrophobic).
The membrane is selectively permeable, allowing some substances to pass while blocking others. Small, nonpolar molecules (O2, CO2, ethanol) can pass through easily. Large polar molecules (glucose) and ions (Na+, K+) require assistance.
Oxygen and nitrogen are highly electronegative, causing them to pull electrons toward themselves in a covalent bond. This creates a polar molecule with partial positive and negative charges.
Proteins
Amino acids can link together to form:
- Peptides (2-9 amino acids)
- Polypeptides (10-100 amino acids)
- Proteins (>100 amino acids)
The structure of proteins can be described in four levels:
- 1st structure is the sequence of amino acids
- 2nd structure is folding into α-helixes and β-pleated sheets
- 3rd structure is the overall fibrous or globular shape
- 4th structure is the combination of multiple proteins
There are four forces that fold a polypeptide chain:
- Hydrogen bonds form between polar groups
- van der Waals forces form from temporary dipoles
- Ionic bonds form between charged groups
- Covalent bonds form from sharing electrons
Hemoglobin is dull red when deoxygenated and bright red when oxygenated. This is an example of how conformational changes in proteins can affect their function.
Cell Transport
Molecules can get across the membrane in four ways:
- Diffusion
- Facilitated diffusion (ion channels, carrier proteins)
- Active transport (primary, secondary, vesicular)
Diffusion
The rate of diffusion, or flux, is affected by
- Solubility of molecule ()
- Surface Area ()
- Concentration gradient ()
- Size of molecule ()
- Distance ()
- Temperature
This is given by the equation:
Sometimes the solubility, size of the molecule, and distance are combined into a permeability coefficient ():
Permeability is affected by several factors:
- Non-polar molecules are more permeable than polar molecules
- Weak acids are permeable when protonated
- Weak bases are permeable when unprotonated
Ion Trapping
Suppose there is a pH gradient across the membrane. Then more weak acids will protonated into on the side that has a higher concentration of . This will increase diffusion of across the membrane. Once inside the cell, the weak acid will dissociate again, trapping it inside.
Note that . That means that a lower pH corresponds to a higher concentration of ions.
Drugs are usually weak acids or weak bases, so they can take advantage of ion trapping. Pharmaceutical companies designing drugs must take into account that the pH of the stomach is between 1-2, and the pH of the small intestine is between 6-7.
Facilitated Diffusion
Facilitated diffusion can happen through ion channels and carrier proteins.
Ion channels have three properties: selectivity, permeability, and gating. Outside of the cell, each ion is surrounded by water molecules (sphere of hydration), which gives the ion some geometry. The aqueous pore of the ion channel has the same geometry as the sphere of hydration, allowing the ion to pass through.
Carrier proteins have three properties: specificity, saturability and competition. Saturability and competition arise because there are a limited number of carrier proteins and potentially multiple types of molecules that can bind to them.
Here are the intracellular and extracellular concentrations of ions:
| Ion | Intracellular (mM) | Extracellular (mM) |
|---|---|---|
| Na+ | 15 | 142 |
| K+ | 150 | 4 |
| Cl- | 5 | 120 |
| Ca++ | 0.0001 | 1 |
Active Transport
Primary active transport uses ATP to move molecules against their concentration gradient.
An example is the sodium-potassium pump (Na+/K+-ATPase), which moves 3 Na+ ions out of the cell and 2 K+ ions into the cell for each ATP hydrolyzed.
Secondary active transport uses the energy from one molecule moving down its concentration gradient to move another molecule against its concentration gradient. This can be done via cotransport (same direction) or countertransport (opposite direction).
For example, the sodium–glucose cotransporter (SGLT) uses the energy of Na+ moving down its electrochemical gradient to transport glucose into the cell against its concentration gradient. This process is driven in part by the negative resting membrane potential.
Vesicular transport uses vesicles to move large molecules across the membrane. Endocytosis brings molecules into the cell, while exocytosis sends molecules out of the cell.
Osmosis and Tonicity
Osmosis
Osmosis is the diffusion of water across a selectively permeable membrane (through aquaporin channels).
Osmotic pressure () is a measure of how strongly water wants to move into a solution. If one side of the membrane has more solute particles (and higher osmotic pressure), then water will move toward that side.
The equation for osmotic pressure is given by:
Where:
- is the concentration of solute particles in mOsm/L
- is the temperature in K
- is the gas constant in atm/mol · K
Note that mOsm is the number of particles a substance dissolves into in the solution. For example, NaCl dissolves into Na+ and Cl-, so 1 mole of NaCl produces 2 Osm.
Tonicity
Tonicity is a measure of how a solution affects cell volume.
- Hypertonic solutions cause cells to shrink
- Isotonic solutions cause no change in cell volume
- Hypotonic solutions cause cells to swell
For human cells, an isotonic solution is generally 300 mOsm/L.
Note that tonicity is only affected by the concentration of non-penetrating solutes. There is also a measure called osmolarity that depends on the total concentration of solutes (both penetrating and non-penetrating).
Filtration and Absorption
Fluid moves from the capillaries into the interstitial fluid (filtration), and from the interstitial fluid back into the capillaries (absorption). To do this, it must pass through the endothelial cells that line the capillaries. This can happen through diffusion, bulk flow, or vesicular transport.
Fluid exchange between capillaries (plasma) and interstitial fluid is governed by Starling forces:
- Hydrostatic pressure caused by the pumping action of the heart pushes fluid out of the capillaries. This pressure decreases along the length of the capillary.
- Colloid osmotic pressure from the proteins within the capillary pulls fluid into the capillaries. It remains relatively constant along the length of the capillary.
This is why filtration generally occurs at the arterial end of the capillary (higher hydrostatic pressure) and absorption occurs at the venous end of the capillary (lower hydrostatic pressure).
Starling forces also explains why elevating an injured limb helps reduce swelling. It decreases hydrostatic pressure, reducing fluid leakage into the interstitial space.
Recently, the glycocalyx layer lining the endothelial cells has been found to play an important role in fluid exchange. It acts as a barrier to protein movement, modulating colloid osmotic pressure across the capilary. This promotes more filtration and less absorption.

Cystic Fibrosis
Cystic fibrosis is a genetic disorder caused by mutations in the CFTR gene, which encodes a chloride channel protein. Cystic fibrosis causes the chloride channel to be malformed and marked by degradation, preventing it from reaching the cell membrane.
Chloride is trapped inside of the cell, preventing water from moving outside to thin the mucus. Chloride is also not reabsorbed, resulting in very salty sweat.
In the pancrease, the lack of chloride secretion limits Na+ secretion, which then limits water secretion, causing pancreatic exocrine secretion to be too thick. Finally, this causes digestive enzymes to not be secreted into the GI track.
Some treatments for cystic fibrosis include:
- Drugs that chaperone CTFR to the membrane to prevent its premature degradation
- Drugs that increase conductance of Fl
Cell Signaling
Chemical communication occurs in the nervous system and the endocrine system.
Steps:
- Ligand released
- Ligand binds to receptor
- Receptor protein have conformational change
Receptors differ in the number of ligands they can bind to (specificity) and their affinity for those ligands.
If there are multiple types of ligands that can bind to the same receptor, then they will compete for binding. Some ligands are agonists, which activate the receptor, while others are antagonists, which block the receptor. Receptors can become saturated when all of the receptors are occupied by ligands.

There are serveral types of receptor responses:
- Ligand-gated ion channels open or close
- Tyrosine kinase have enzymatic activity
- Receptors interact with cytoplasmic janus kinases
- G-protein coupled receptors activate
G-proteins contain an alpha, beta, and gamma subunit. When the first messenger binds to the receptor, it increases the affinity of the alpha subunit for guanosine triphosphate (GTP). When bound to GTP, the alpha subunit dissociates and links up with an effector protein in the membrane. Once it activates the protein, the GTP is cleaved back into GDP, allowing the alpha subunit to recombine with the G-protein group.
When adenylyl cyclase is activated by the alpha subunit, it takes in ATP and creates cyclic adenosine monophosphate (cAMP). cAMP acts as a second messenger and binds to an enzyme known as cAMP-dependent protein kinase (protein kinase A). Eventually, cAMP terminates when it is broken down to AMP.
Question: Why is this process so complicated?
The benefits of having a multistep process is (1) amplification, (2) modulation, and (3) duration.

Question: How can the same second messenger cause two different cellular responses?
Each receptor is surrounded by different effector proteins, protein kinases, and phosphatases. This allows the same second messenger to cause different responses in different compartments.
Endocrine System
The hypothalamus produces releasing factors that are released into the capillary bed in the pituitary stalk. They stimulate the anterior pituitary gland, causing it to release hormones through the bloodstream. These hormones can have direct effects on target tissues or stimulate other endocrine glands to release hormones.
For example, the hypothalamus releases thyrotropin-releasing hormone (TRH), which stimulates the anterior pituitary to release thyroid-stimulating hormone (TSH). TSH then stimulates the thyroid gland to release thyroid hormones (T3 and T4) in the bloodstream. T3 and T4 are lipid-soluble hormones that enter cells and bind to nuclear receptors, affecting gene transcription.
Hyperthyroidism causes increased metabolic rate and body temperature.
Question: How do you store something that is lipid-soluble?
T3 and T4 are covalently bonded together in a non-penentrating protein called thyroglobulin and stored in the colloid of the thyroid gland. When T3 and T4 are needed, the cell endsocytoses the thyroglobulin, breaks the covalent bond, and releases T3 and T4 into the bloodstream.
Question: What is determining the volume of the thyroid gland?
Most of the volume of the thyroid gland comes from the colloid. When the colloid swells up, the thyroid gland swells up and becomes a goiter. The colloid swells up when there is high osmotic pressure (caused by a high concentration of thyroglobulin).
Question: What happens when...
| Condition | TSH | T3 & T4 | Goiter |
|---|---|---|---|
| Hyperactive thyroid stimulation | Low | High | Yes |
| Hyperactive hypothalamus or anterior pituitary | High | High | Yes |
| Hypersecreting thyroid | Low | High | No |
| Thyroid failure or low iodine | High | Low | Yes |
| Inactive hypothalamus or anterior pituitary | Low | Low | No |
Neurons, Synapses, and Signaling
Membrane Potential
All cells exhibit a membrane potential (Vm) across the membrane. The inside of the cell is typically negative relative to the outside. This potential energy can be used to do work. Excitable cells (neurons, muscles) use changes in membrane potential to send signals.
Cells use energy (ATP) to create a chemical gradient with K+ and Na+. K+ has a higher concentration inside the cell, while Na+ has a higher concentration outside the cell.
If more K+ channels are opened, then more K+ will leave the cell, making the inside more negative. If more Na+ channels are opened, then more Na+ will enter the cell, making the inside more positive. Ion channels will not appreciably change the concentration of K+ and Na+.
Note that 3 Na+ are pumped out of the cell and 2 K+ are pumped into the cell for each ATP hydrolyzed, so there is also a small membrane potential from that.
If there are multiple permeable ions, then the membrane potential is given by the Goldman-Hodgkin-Katz equation:
Where:
- is the permeability of the ion, which is impacted by number of ion channels
- is the concentration of the ion
Equilibrium potential
To reach electrochemical equilibrium, the chemical gradient must be balanced by the electrical gradient. Relatively few ions need to move to create a significant electrical gradient. This is described by the Nernst equation:
Where:
- is the gas constant
- is the temperature
- is the ion charge
- is Faraday's constant
Current
From Ohm's Law, we can derive this equation for current:
Where:
- is current
- is the membrane conductance
- is the membrane potential
- is the equilibrium potential
Note that:
- When is positive, there is an outward current (positive charge exiting the cell or negative charge entering the cell).
- is maintained at a constant level.
- changes rapidly.
- has some components that are constant (leak channels) and some components that change rapidly (gated channels). can change rapidly.
- Driving force is the difference between the membrane potential and the equilibrium potential .
If the number of open ion channels changes, then the conductance changes, which changes the current . This causes more particles to either enter or leave the cell, which changes membrane potential . Membrane potential asymptote toward equilibrium potential . At steady-state, the net current should be zero.
If there is a delayed rectifier potassium channel, the membrane potential may undershoot the equilibrium potential.
Neurons
Neurons are very large cells with high metabolic demands. As soon as resources are cut off, they immediately start to malfunction. Long projecting neurons are the first to show signs of disease, which manifests as tingling or numbness in hands in feet.
Astrocytes line the capillaries and tightly regulate what can come out of it (blood-brain barrier).
Afferent neurons receive signals from sensory receptors. Interneurons in the central nervous system connect the afferent neurons to the efferent neurons. The efferent neuron sends signals to muscles, glands, and other tissue through the axon terminal.
Neuron anatomy:
- Dendrites receive inputs from other cells
- Soma (cell body) integrates all input
- Axon hillock decides whether to fire an action potential
- Axon conducts the action potential down the length of the neuron
- Presynaptic axon terminals release chemical signalling molecules
Action Potential
Neurons communicate with each other through action potentials and neurotransmitters.
- An action potential arrives at the presynaptic axon terminal.
- The depolarization causes voltage-gated calcium channels to open.
- Ca2+ enters the cell, causing exocytosis of synaptic vesicles containing neurotransmitters.
- The neurotransmitters diffuse across the synaptic cleft and bind to receptors on the postsynaptic cell.

Voltage-gated sodium channels have three states: closed, open, and inactivated. When depolarizing stimulus brings the membrane to a threshold potential, the voltage-gated sodium channels rapidly open, allowing Na⁺ to enter the cell and further depolarize the membrane. This depolarization triggers additional sodium channels to open through positive feedback, creating the action potential. Shortly after opening, the channel becomes inactivated, preventing further Na⁺ influx even if the membrane remains depolarized. The channel cannot reopen until the membrane repolarizes toward its resting potential, at which point it returns to the closed state.
Delayed rectifier potassium channels are voltage-gated channels that open in response to depolarization. As the voltage-gated sodium channels are inactivated, the potassium channels open up, allowing the membrane to return back toward its resting potential. This is an example of a negative feedback loop.

Conduction and Synapses
Graded potentials can only travel a short distance before diminishing in amplitude. To send signals beyond a very short distance, action potentials must be generated and propagated along the entire length of neuron.
Current enters the cell and has three pathways:
- Down the inside of the axon ()
- Through the cell membrane ()
- To the cell membrane to charge up the capacitance ()
We would like current to travel down the axon, but the problem is that axons are poor conductors ( is high), and pathways (2) and (3) are significant drains on overall current.
These problems are solved by several adaptations:
- Having larger axon diameters, decreasing
- Having no open ion channels between nodes, increasing
- Having a thick myelin sheath, decreasing
Every so often, there are gaps in the myelin sheath called nodes of Ranvier where the voltage-gated sodium channels are concentrated. These regions are where the action potential is regenerated.
Local anesthetic works by blocking action potentials in small diameter axons (like those that handle pain). It spares large diameter axons like touch and motor.
Nervous System
Types of Nervous Systems
The central nervous system (CNS) is comprised of the brain and spinal cord.
- The hypothalamus controls the pituitary hormones.
- The medulla controls breathing and heart rate.
The peripheral nervous system (PNS) has three components:
- The somatic controls voluntary movements by skeletal muscles.
- The autonomic controls involuntary functions by smooth muscles and glands.
- The enteric controls the gastrointestinal system (gut).
The autonomic nervous system can be divided into the sympathetic, which is responsible for the fight-or-flight response, and the parasympathetic, which is responsible for rest-and-digest functions.
Activation Pathways
In the somatic nervous system, the axon releases acetylcholine (ACh), which binds to nicotinic ACh receptors on the skeletal muscle, causing it to contract.
In the automatic nervous system, preganglionic neurons releases ACh, which binds to nicotinic ACh receptors on the postganglionic neuron. The postganglionic then releases a different neurotransmitter depending on whether it is part of the sympathetic or parasympathetic nervous system.
In the sympathetic nervous system, the postganglionic neuron then releases norepinephrine, which binds to adrenergic receptors on the target tissue. The adrenergic receptors can be either alpha or beta, and they can have different effects on the target tissue. Alpha-1 adrenergic receptors cause vasoconstriction, while beta-2 adrenergic receptors cause vasodilation.
In the parasympathetic nervous system, the postganglionic neuron then releases ACh, which binds to muscarinic ACh receptors on the target tissue
Visceral and Somatic Motor System
Muscles are made of muscle fibers, which contain myofibrils composed of repeating sarcomeres. The sarcomere is the basic contractile unit of muscle and contains thin filaments (actin) anchored to the zig-zagging Z lines and thick filaments (myosin) anchored at the middle M line. The actin contains regulatory proteins (troponin and tropomyosin) which control whether it can bind to myosin.

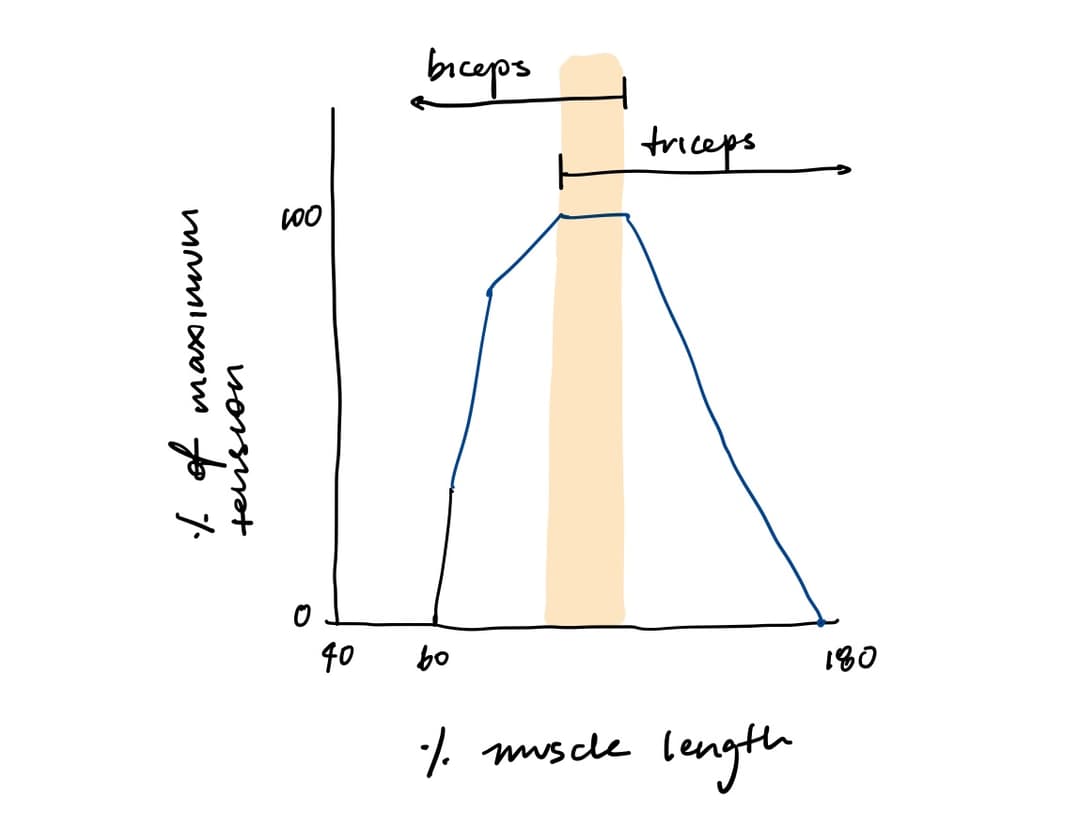
Sarcomeres have an optimal length at which they can generate the most force. If they are stretched too much, then there will be less overlap between actin and myosin, so fewer cross-bridges can form. If they are stretched too little, then actin filaments from one side interfere with crossbridge formation on the other side. This is called the length-tension relationship.
For example, when the arm is extended, both biceps and triceps have optimal overlay. When the arm is flexed, biceps are shorter than optimal and triceps are longer than optimal.
The neuromuscular junction is the synapse between a motor neuron and a skeletal muscle fiber. The motor neuron releases ACh, which binds to nicotinic ACh receptors on the muscle fiber, causing depolarization of the sarcolemma and propagation of an action potential.
T-tubules conduct the action potentials, allowing them to reach the interior of the cell. In the T-tubules, depolarization activates dihydropyridine (DHP) receptors. They are mechanically coupled to ryanodine receptors on the sarcoplasmic reticulum, so their conformational change causes Ca++ to release from the sarcoplasmic reticulum.
Calcium binds to troponin, causing tropomyosin to shift away from myosin-binding sites on actin. A crossbridge forms between the myosin heads and the actin when myosin has ADP and Pi bound and troponin has Ca++ bound. This causes muscle contraction.
Muscular force is correlated with (1) the concentration of intracellular calcium as well (2) the number of cross-bridges per second.
Cardiac System
The heart is a dual pump:
- The pulmonary side pumps blood through the pulmonary artery to the lungs
- The systemic side pumps blood through the aorta to the rest of the body
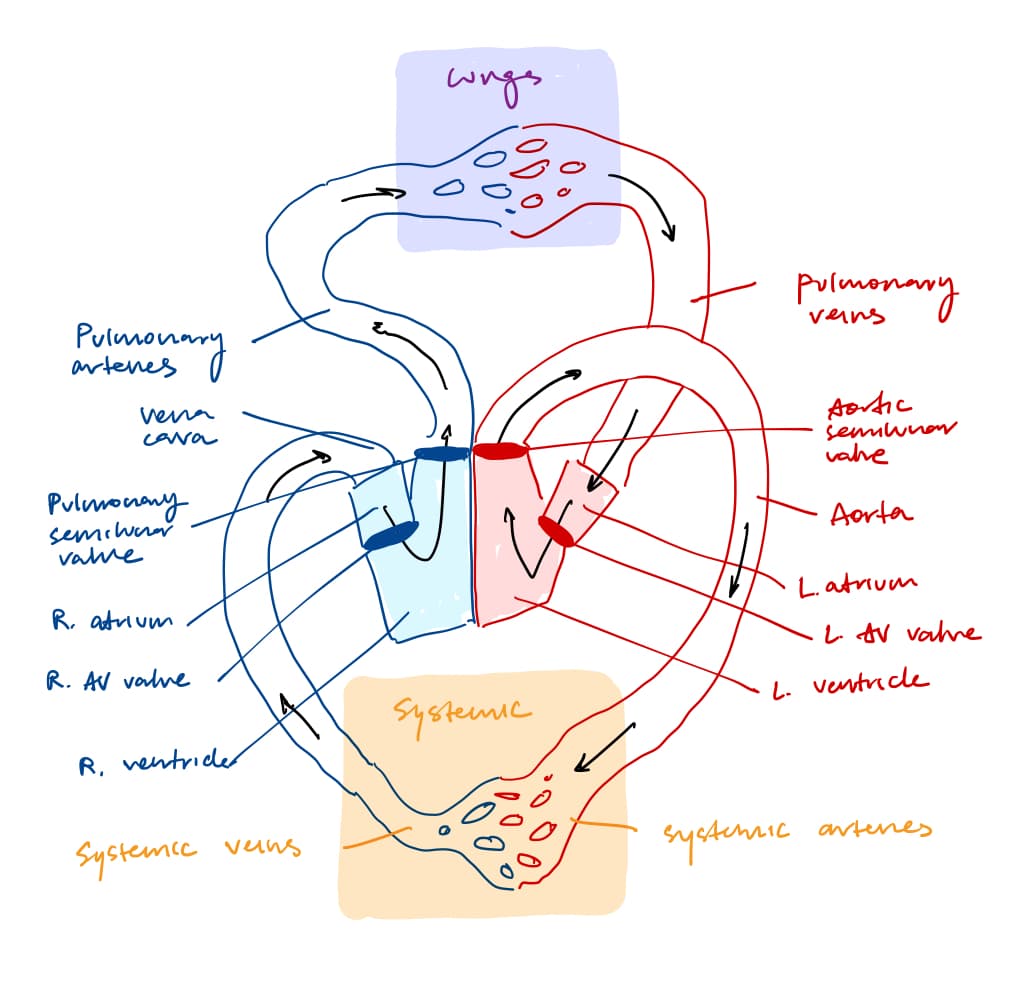
The atrium receives blood moving into the heart. The ventricle receives blood from the atrium.
Valves open and close depending on pressure in the two chambers. Valves can be blown open (prolapse) if the pressure difference is too high, leading to backflow of blood. Capillary muscles hold the valve in place to prevent a prolapse.
There are multiple valves in the heart:
- The atrium-ventricular (AV) valve separates atrium and ventricle
- The aortic and pulmonary semilunar valves separate the ventricles from the aorta and pulmonary artery
Usually, the first sound of the heartbeat is caused by the closing of the AV valves, and the second sound is caused by the closing of the semilunar valves. Murmurs can be caused by valves not opening well (stenoic) or not closing well (insufficient).
Arteries bring blood away from the heart and veins bring blood toward the blood. Capillary beds are where the blood becomes deoxygenated and picks up CO2.
Cardiac Excitation
Pacemaker cells in the sinoatrial (SA) node automatically generate heartbeats and pass signals to contractile muscle cells through gap junctions.
- When the membrane is very negative, funny NA+ channels open; Na+ enter the cell
- Voltage-gated T-type Ca2+ channels to open briefly; Ca2+ enter the cell
- At threshold, L-type Ca2+ channels open; more Ca2+ enter the cell
- The action potentials move to the contractile cells, causing them to contract and pump blood
The SA node is the main pacemaker and operates at 60-100 bpm. The atrioventricular (AV) node can also act as a pacemaker, but it generates action potentials at a slower rate of 40-60 bpm. If both the SA and AV nodes fail, the His bundle can generate action potentials at an even slower rate of 20-40 beats per minute.
From the SA node, the action potential travels from the atria to the AV nodes to the His bundle to to the ventricles.
Ventricular action potential:
- Rapid depolarization: voltage-gated Na+ channels open; Na+ enter the cell
- Brief repolarization: voltage-gated Na+ channels inactivate; some K+ channels open; K+ exit the cell
- Plateau: voltage-gated Ca2+ channels open; Ca2+ enter the cell and balance the outward K+ current, creating a plateau in membrane potential
- Repolarization: voltage-gated Ca2+ channels inactivate, and more K+ channels open, allowing K+ to exit the cell and repolarize the membrane back to its resting potential
EKG
A EKG consists of a few phases:
- P wave: depolarization of the atria
- PR interval: conduction delay in the AV node
- QRS complex: depolarization of the ventricles
- T wave: repolarization of the ventricles
Cardiac Cycle
In a cardiac cycle, there is first isovolumetric relaxation, where both valves are closed as pressure rises. Then the AV valves open, the semilunar valves close, and blood fills the ventricles (diastole). Then there is isovolumetric contraction again, where both valves are closed as pressure rises. Then the AV valves close, the semilunar valves open, and blood is ejected (systole).
| Term | Definition |
|---|---|
| Diastole | Muscle relaxation in ventricles |
| Systole | Muscle contraction in ventricles |
| Isovolumetric contraction | Period where both valves are closed as pressure rises |
| End diastolic volume | Volume of blood in ventricles at the end of diastole (~120 mL) |
| End systolic volume | Volume of blood in ventricles at the end of systole (~50 mL) |
| Stroke volume | Amount of blood ejected from ventricles (EDV - ESV) |
| Ejection fraction | Percentage of blood ejected from ventricles (SV/ EDV) |
| Preload | End-diastolic volume |
| Afterload | Resistance ventricles must overcome to eject blood |
Frank-Starling Mechanism
The Frank-Starling mechanism states that the more the ventricles are filled during diastole, the more they will contract during systole.
Question: There is no change in sympathetic and parasympathetic tones, so it must be an intrinsic property of the heart. How does it work?
Sarcomeres can generate the most force when they are stretched to 2.0-2.2 micrometers. When the ventricles are filled more, the sarcomeres are stretched closer to their optimal length, allowing for more cross-bridges to form and a stronger contraction.
The problem is that the length-tension effect only accounts for a small increase in force. There are two additional mechanisms that contribute to the Frank-Starling mechanism:
- Stretch-sensitive calcium channels let more calcium into the cell, allowing more cross-bridges to form.
- Stretch-sensitive troponin binds more strongly to calcium, allowing more cross-bridges to form.
Increased sympathetic stimulation can also cause increased calcium concentration and increased force.
Pressure-Volume Loops
The PV loop represents the changes in pressure and volume in the ventricles during a single cardiac cycle.
There are four phases of the PV loop:
- Isovolumetric relaxation
- Diastolic filling
- Isovolumetric contraction
- Systolic ejection
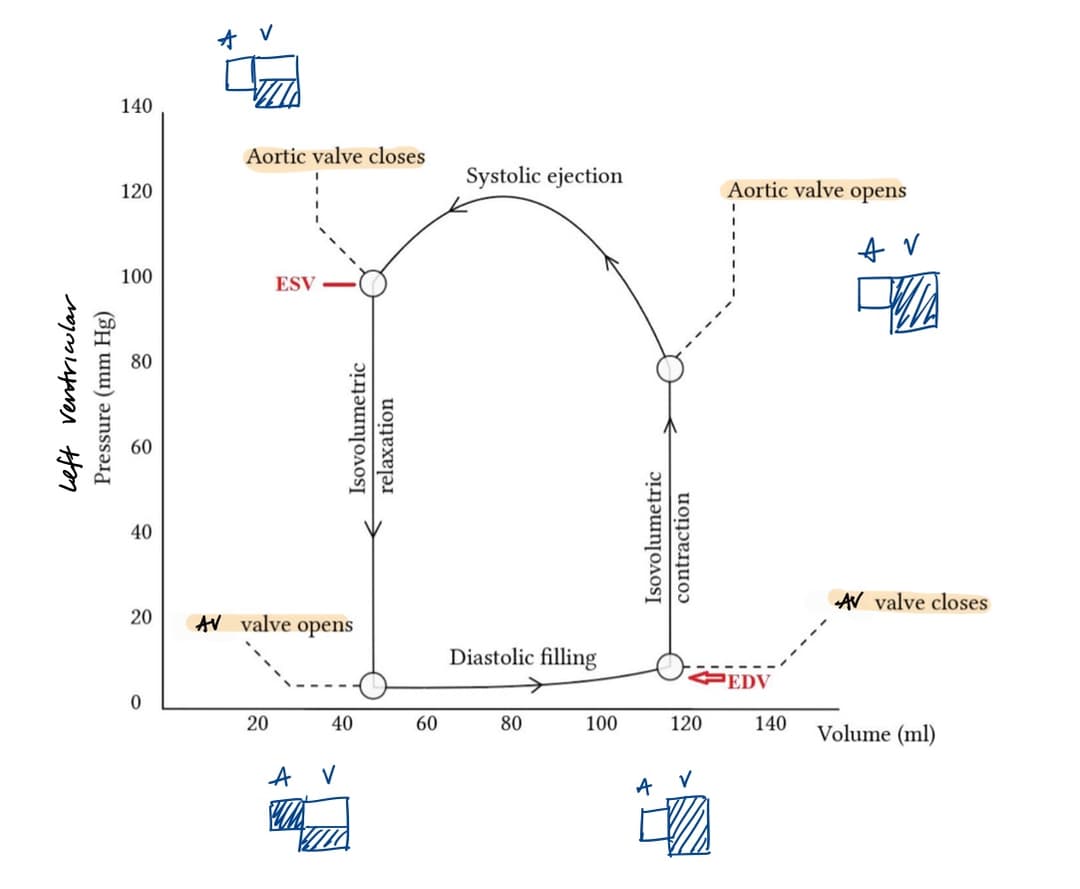
The diastolic pressure curve represents the passive filling of the ventricle and is impacted by how easily the ventricle stretches (compliance).
The systolic pressure curve represents the resistance the ventricle must overcome to eject blood and is impacted by how strongly the ventricle contracts (contractility).
Contractility is influenced by the Frank-Starling mechanism, which is affected by preload, afterload, and sympathetic tone.
The actual preload is determined by the venous return, and the actual afterload is determined by the arterial pressure and TPR.
Stroke volume is impacted by preload, afterload, and sympathetic tone. If preload is higher, then the Frank-Starling mechanism kicks in—resulting in higher stroke volume. If afterload is lower, then more work can go into ejecting volume rather than generating higher pressure—resulting in an increase in stroke volume.
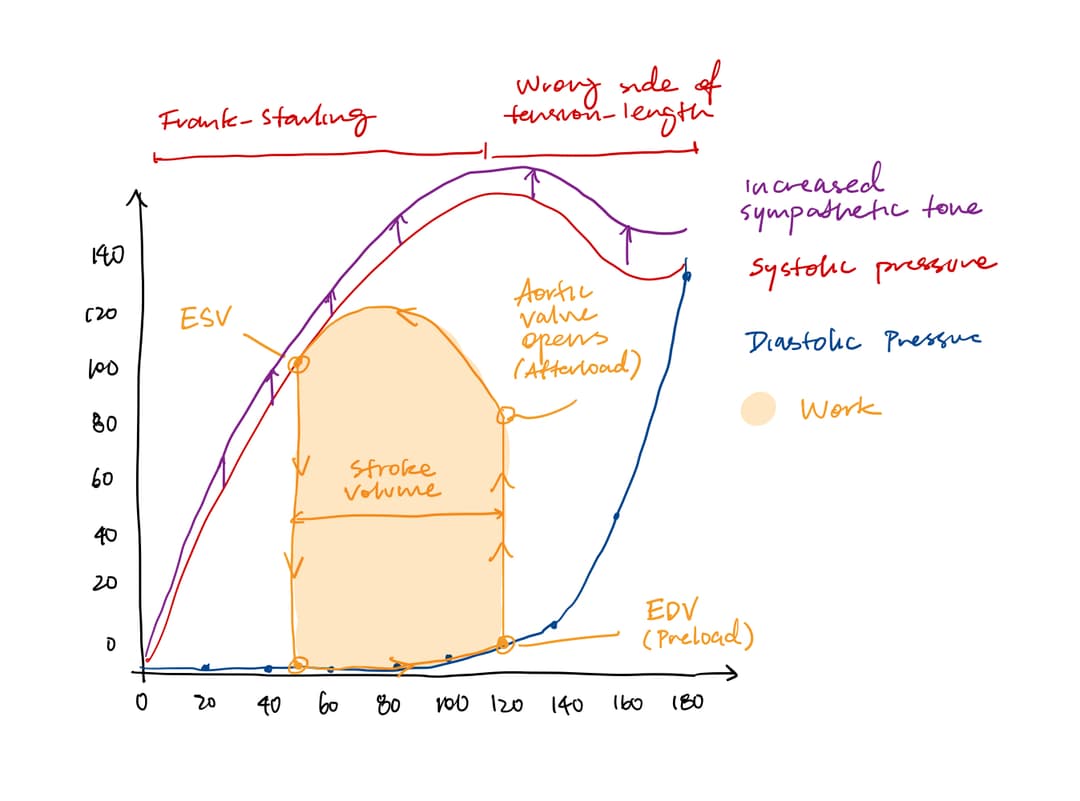
Wiggers Diagram
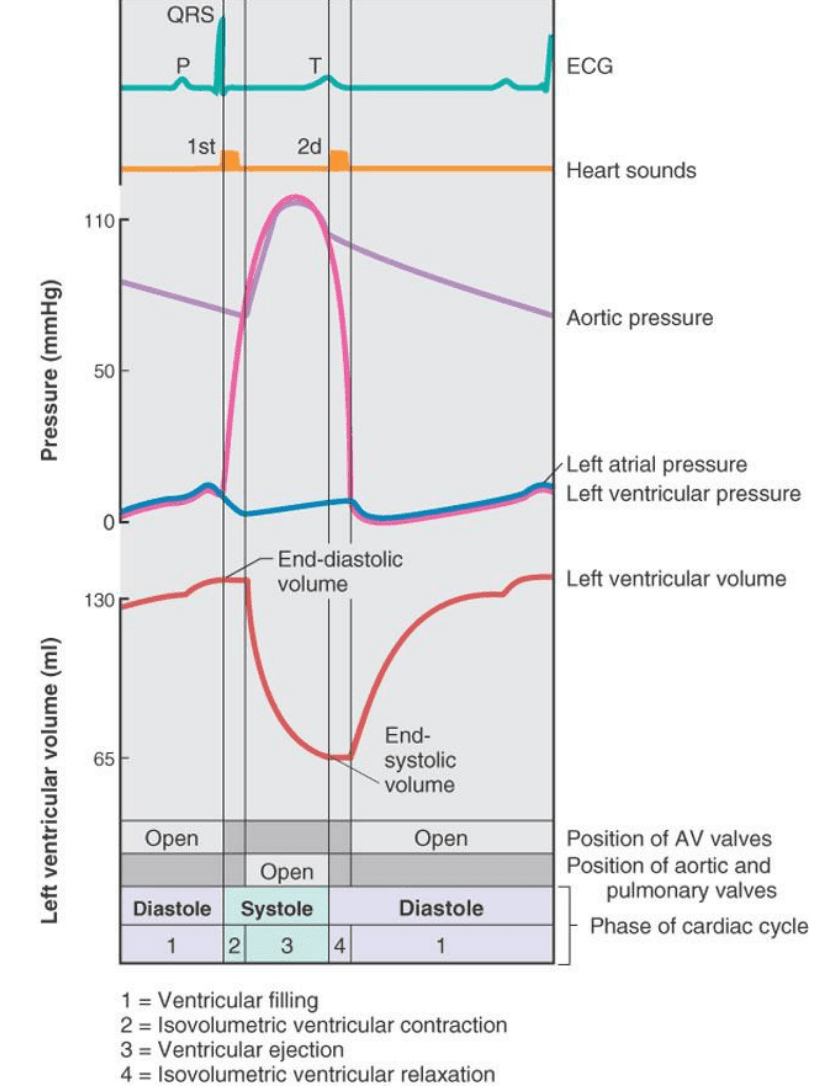
Notes:
- Whenever the pressure curves cross, a valve changes state
- Most filling of the ventricles is passive
- Most filling ocurrs in the first third of diastole
- Most emptying occurs in the first third of systole
Blood Flow
Blood Cells
Blood cells don't have a nucleus, so they can't make new proteins. They have a lifespan of around 120 days, after which they are removed by the spleen and replaced by new blood cells made in the bone marrow.
RC Circuit
Blood flow is similar to an RC circuit. The small blood vessels have high resistance. The arteries have low resistance and act as a capacitor for pressure.
As you age, your arteries become stiffer, so they are less able to stretch and recoil. This causes a higher systolic pressure and a lower diastolic pressure, leading to a higher pulse pressure.
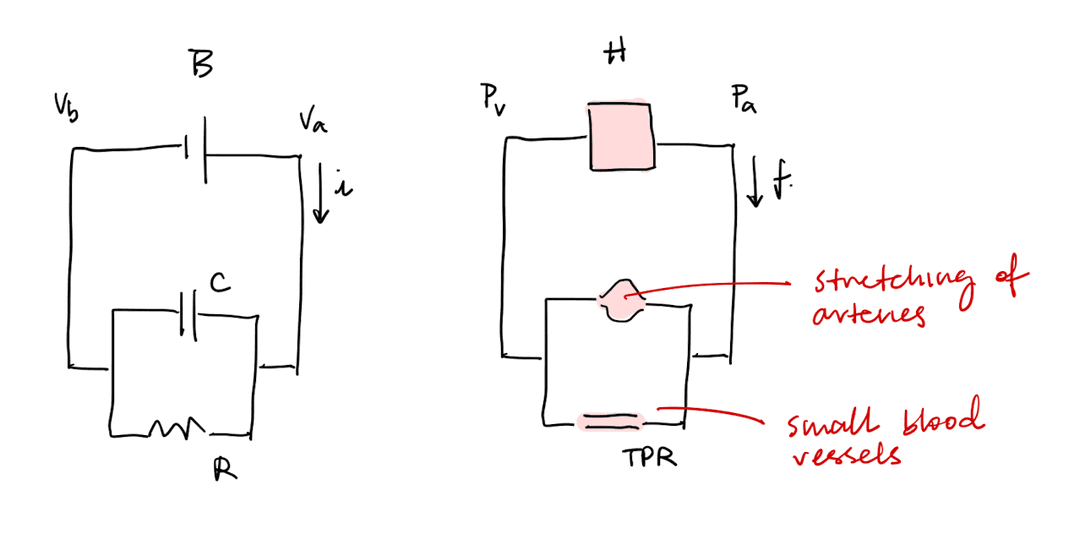
Increased capacitance of the veins decreases venous return, decreasing preload and thus stroke volume. Increase capacitance of arteries decreases the amount that pressure rises when the heart ejects blood (pulse pressure). Note that veins tend to have greater capacitance than wrteries.
Capacitance of the veins and arteries does not affect total peripheral resistance, which is determined by the arterioles.
Blood Pressure
The equation for blood pressure is
Where:
- is arterial pressure
- is venous pressure, which is almost
- CO is cardiac output
- TPR is total peripheral resistance
The equation for cardiac output is
Where:
- CO is cardiac output (mL/min)
- HR is heart rate (beats/min)
- SV is stroke volume (mL/beat)
Heart rate in the SA node is determined by the degree of sympathetic and parasympathetic stimulation. Stroke volume is determined by contractility and the Frank-Starling mechanism, which is influenced by preload afterload, and sympathetic tone. One tricky part is that affects the afterload, which then affects stroke volume. When increases, the afterload increases, which decreases stroke volume.
Total peripheral resistance is the overall resistances in the blood vessels. Resistance of the blood vessels is determined by Pouseille's Law, which states
Where:
- is resistance
- is the viscosity of the fluid
- is the radius of the vessel
TPR is only affected by vasodilation and vasoconstriction, which is impacted by sympathetic and parasympathetic tone.
Pressure Gradient
Blood pressure is highest in the aorta and decreases as it moves through the arteries, arterioles, capillaries, venules, veins, and back to the heart. The largest drop in pressure occurs in the arterioles, which are the main site of resistance in the circulatory system.
Average aortic pressure is diastolic pressure plus one-third of the pulse pressure, or around 100 mmHg.
Question: Blood flows out of the heart because there is a standing pressure gradient. But there is no pressure gradient to drive blood flow in the veins. How does blood flow back to the heart?
There is a lot of blood contained in chambers separated by valves. When joints and muscles push on the walls of these chambers, pressure increases, forcing blood to flow through the one-way valves back to the heart.
Actually, if you stand still for a very long time, there may not be enough blood flowing back to the heart, causing you to faint.
Sympathetic and Parasympathetic Tone
The brain monitors and maintains blood pressure through baroreceptors in the carotid sinus and aortic arch. When their action potential firing decreases, the brain increases sympathetic and decreases parasympathetic activity to raise blood pressure.
For example, during a valsalva maneuver, you exhale forcefully against a closed airway, which increases pressure in the chest and decreases venous return. This causes a decrease in stroke volume and cardiac output, which causes a drop in blood pressure. The baroreceptors detect this drop in blood pressure and increase sympathetic tone to compensate.
Effects of sympathetic tone:
- Contractility increases, causing the heart to beat faster and stronger. This increases stroke volume and cardiac output.
- Vasoconstriction increases, causing total peripheral resistance to increase. This helps maintain blood pressure even when cardiac output increases.
- Veins stiffen, decreasing capacitance. This causes a greater increase in pressure when the muscles compress the veins, resulting in a greater venous return. As a result, preload increases, which increases stroke volume and cardiac output.
Specifically, the sympathetic system uses norepinephrine to bind to alpha-1 adrenergic receptors on the blood vessels, causing vasoconstriction and increasing TPR. It also uses norepinephrine to bind to beta-1 adrenergic receptors on the heart, increasing heart rate and contractility, which increases cardiac output.
The parasympathetic system uses ACh to bind to muscarinic receptors on the heart, decreasing heart rate and contractility, which decreases cardiac output.
Vasodilation and Vasoconstriction
Smooth muscle and precapillary sphincter muscles are found in the walls of blood vessels. They can contract or relax to change the radius of the vessel, which changes resistance and blood flow.
Precapillary sphincter muscles regulate their own oxygen supply. When they constrict, less blood flows to local capillaries and oxygen levels drop. When oxygen levels drop, they can no longer manufacture ATP at the same rate, causing them to become weak and relaxed.
The strength of the precapillary sphincter muscles is determined by the number of crossbridges formed, which is affected by the concentration of calcium and the ATP.
The following factors can cause dilation:
- Decreased oxygen levels
- Increased temperature
- Increased carbon dioxide levels
- Decreased pH
- Increased adenosine levels
Multiple factors can affect blood flow:
- Precapillary sphincter muscles
- Upstream artery effect through nitric oxide
- Hormones in the blood such as histamine, epinephrine, antidiuretic hormone (ADH) and angiotensin
Vasodilation in the precapillary sphincter muscles causes an increase in blood flow, which increases shear forces on the upstream arteries. This causes an increase in nitric oxide production, which causes vasodilation in the upstream arteries. This is called the upstream artery effect.
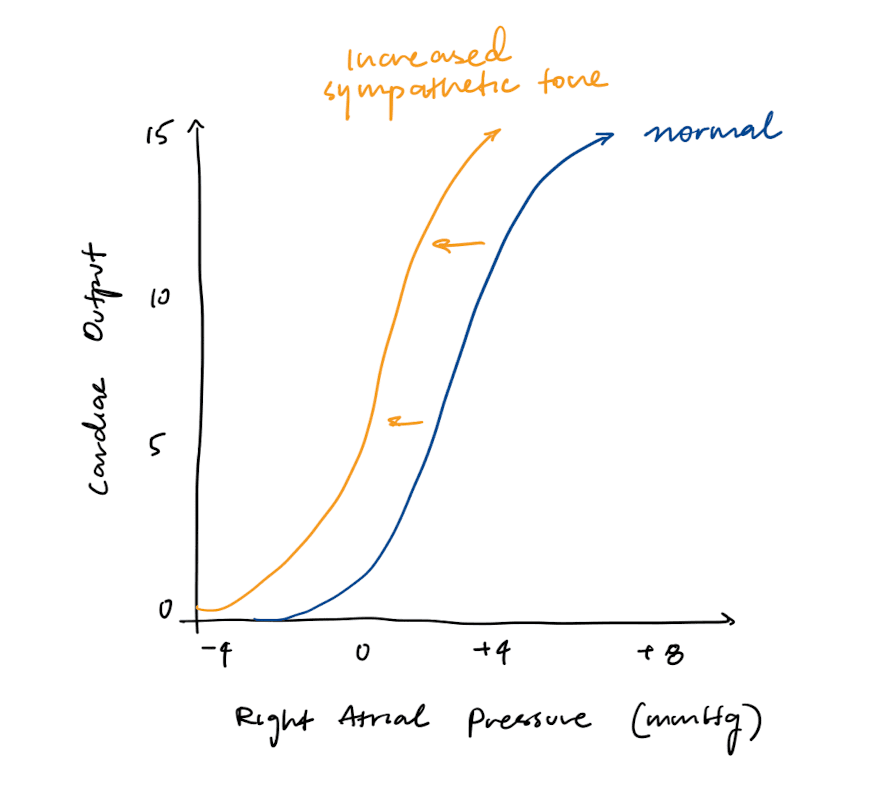
Cardiac Function Curve
The cardiac function curve shows that as right atrial pressure (RAP) increases, there is more filling (preload), increasing cardiac output.
High right atrial pressure implies that there is a high venous return. Assuming that the right heart pushes blood effectively through the pulmonary system to the left heart, this means that there is high preload in the left ventricles as well. High preload causes increased stroke volume and thus increased cardiac output.
There are two things that can affect the cardiac function curve:
- Heart rate
- Contractility
Increasing sympathetic tone increases both heart rate and contractility, so it will shift the cardiac function curve to the left.
Vascular Function Curve
The vascular function curve shows that as right atrial pressure (RAP) increases, there is less of a pressure gradient, decreasing venous return.
Blood flow between the veins and the right atrium depends on the pressure gradient between them. When right atrial pressure is high, there is a smaller pressure gradient, so blood flows back to the heart slowly, decreasing venous return.
There are three things that can affect the vascular function curve:
- Blood volume
- Capacitance of veins
- Total peripheral resistance.
If blood volume or capacitance of veins increases, then the venous return will increase at a given right atrial pressure, shifting the vascular function curve to the right. This moves the x-intercept to the right, which means that the mean circulatory filling pressure (MCFP) increases.
If total peripheral resistance increases, then the venous return will decrease multiplicatively at every right atrial pressure. The slope of the vascular function curve will decrease.
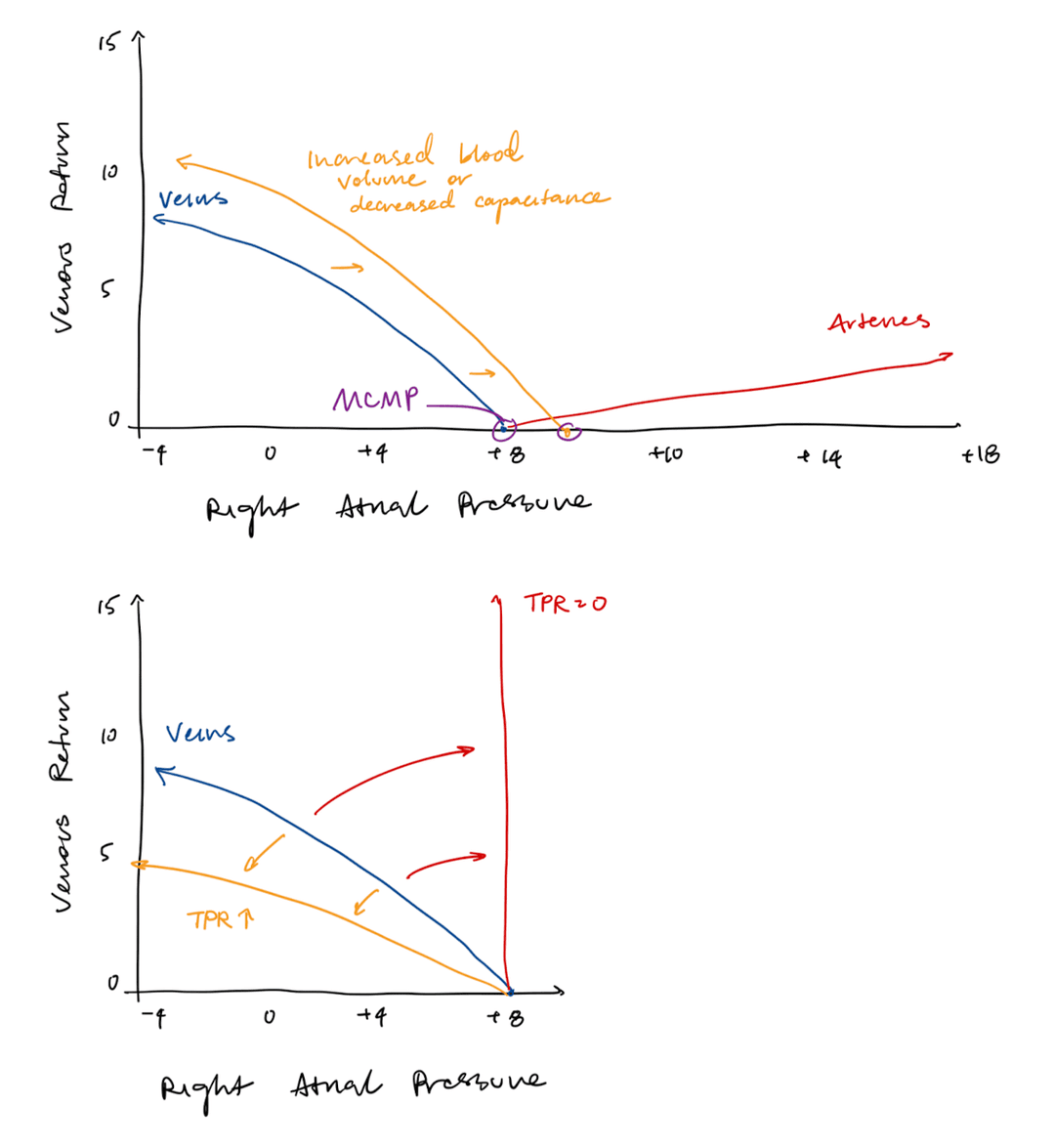
Cardiovascular Curve
At stead state, cardiac output is equal to venous return. Thus, the system should operate at the intersection of the cardio and vascular curves.
Exercise, heart failure, increased blood pressure, and hemorrhage (blood loss) can all affect the cardiac and vascular function curves. To determine how they affect the curves, consider:
- Direct effects
- Effect on blood pressure
- Sympathetic or parasympathetic response
Respiratory System
Structure of the Respiratory System
The respiratory system can be divided into two zones:
- The conducting zone brings in air
- The respiratory zone facilitates gas exchange
The conducting zone walls are lined with smooth muscles, which react to sympathetic and parasympathetic stimulation to eitehr dilate or constrict the airways.
The respiratory zone walls are lined with alveoli, where gas exchange occurs: O2 enters the pulmonary blood and CO2 leaves it.
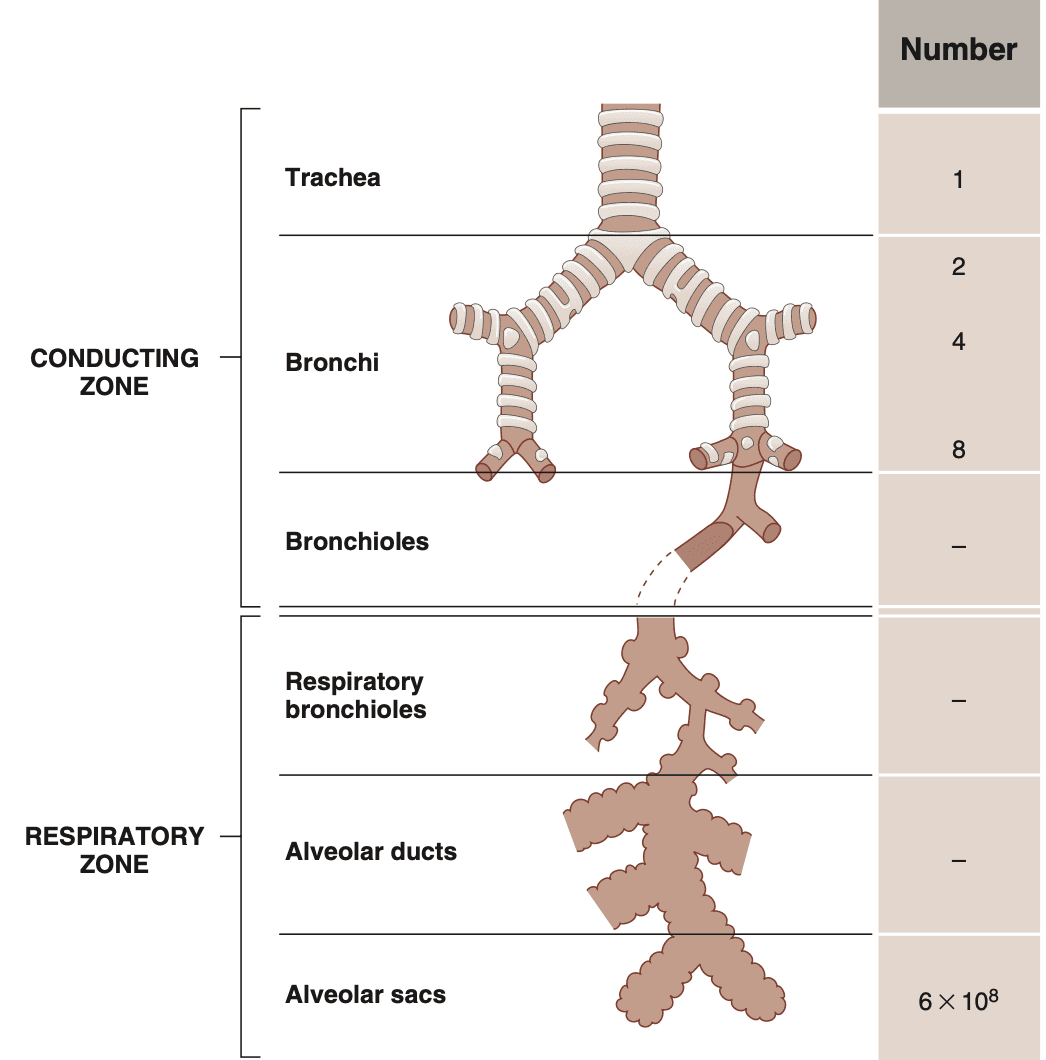
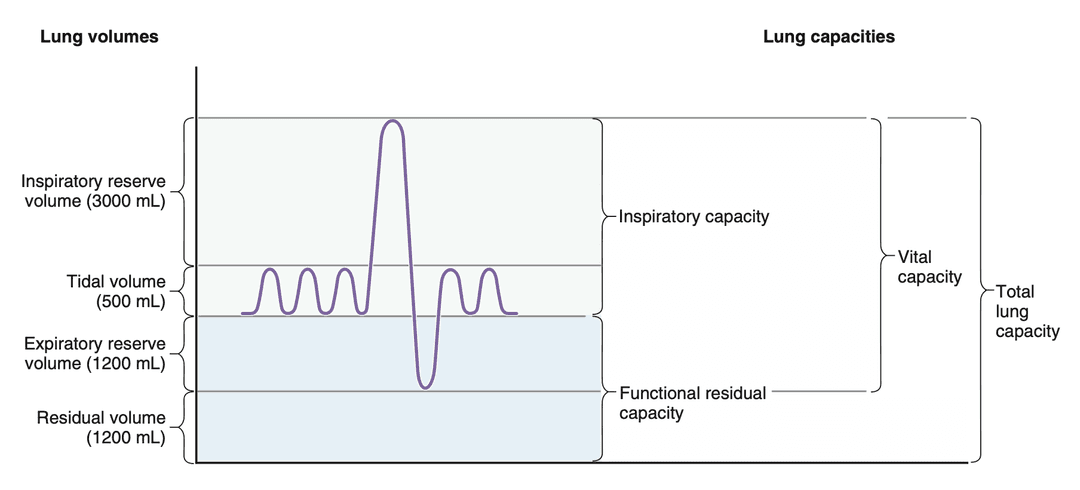
Lung Volume and Capacity
Lung volumes are measured with a spirometer.
First, the subject is asked to breathe normally, in order to measure the tidal volume (TV), which should be around 500 mL. Next, the subject is asked to take a maximal inspiration followed by a maximal expiration. The additional volume that can be inspired above the TV is called the inspiratory reserve volume (IRV), which should be around 3000 mL. The additional volume that can be expired below the TV is called the expiratory reserve volume (ERV), which should be around 1200 mL.
The volume of gas remaining in the lungs after a maximal forced expiration is the residual volume (RV), which is approximately 1200 mL and cannot be measured by the spirometer.
| Term | Definition |
|---|---|
| Tidal volume (TV) | Volume of air inhaled or exhaled during normal breathing |
| Inspiratory reserve volume (IRV) | Additional volume of air that can be inhaled |
| Expiratory reserve volume (ERV) | Additional volume of air that can be exhaled |
| Residual volume (RV) | Volume of air remaining in the lungs after maximum exhalation |
Lung capacities are combinations of lung volumes. For example, the inspiratory capacity (IC) is the maximum volume of air that can be inhaled after a normal exhalation, which is the sum of TV and IRV. The functional residual capacity (FRC) is the volume of air remaining in the lungs after a normal exhalation, which is the sum of ERV and RV. The vital capacity (VC) is the maximum amount of air that can be exhaled, and is composed of the IC plus the ERV. Finally, the total lung capacity (TLC) includes all of the lung volumes.
Because RV cannot be measured by spirometry, FRC and TLC also cannot be measured by spirometry.
| Term | Definition |
|---|---|
| Inspiratory capacity (IC) | Maximum volume of air that can be inhaled |
| Vital capacity (VC) | Maximum amount of air that can be exhaled |
| Functional residual capacity (FRC) | Volume of air remaining in the lung normal exhalation |
| Total lung capacity (TLC) | Maximum amount of air the lungs can hold |
Dead Space
Dead space is the volume of air that does not participate in gas exchange. For example, with a TV of 500 mL, around 150 mL fills the anatomic dead space, and 350 mL fills the alveoli. Dead space air is also the first air expired.
Physiologic dead space can differ from anatomic dead space when there is a defect in the ventilation-perfusion ratio. In that case, it can be calculated this equation:
Where:
- is dead space (mL)
- is tidal volume (mL)
- is arterial partial pressure of CO2
- is expired partial pressure of CO2
In other words, dead space is tidal volume multiplied by some fraction.
Ventilation Rates
Ventilation rate is the rate of air movement in the respiratory system. It can be expressed either as the minute ventilation, which is the rate of air movement into and out of the lungs.
It can also be expressed as alveolar ventilation, which is the rate of air movement into and out of the alveoli. Alveolar ventilation corrects for physiologic dead space.
Where;
- is alveolar ventilation (mL/min)
- is tidal volume (mL)
- is dead space volume (mL)
- is breathing frequency (breaths/min)
Mechanics of Breathing
The lungs tend to collapse inward due to their elasticity. The chest tends to expand outward. The pleural cavity between them maintains a negative pressure, which keeps the lungs inflated and allows these opposing forces to stay in balance.
Surface tension in the alveoli further promotes collapse, but the lungs produce pulmonary surfactant to reduce this tension. By lowering surface tension, surfactant increases lung compliance (capacitance), making it easier for the lungs to expand during breathing.
During inhalation, the diaphragm and intercostal muscles contract, expanding the thoracic cavity and making pleural pressure more negative. This decrease in pressure draws air into the lungs. During exhalation, these muscles relax, the thoracic cavity decreases in size, pleural pressure becomes less negative, and air flows out of the lungs.
In a pneumothorax, air enters the pleural space and disrupts the negative pressure. As a result, the lung collapses inward and can no longer expand properly.
Question: The cellular respiration equation has a 1:1 ratio of oxygen to carbon dioxide. Why do we breathe in more oxygen but breathe out less carbon dioxide?
The 1:1 ratio of O2 to CO2 applies only to glucose metabolism. In reality, the body also metabolizes fats and proteins, which contain less oxygen than carbohydrates. As a result, their oxidation requires more O2 per CO2 produced, so we consume more oxygen than the carbon dioxide we exhale.
This is captured by the respiratory exchange ratio, or the ratio of carbon dioxide produced to oxygen consumed. The average respiratory exchange ratio for a mixed diet is around 0.8.
Lung Conditions
Normal lung. PO2 in the alveoli and blood are around 100 mmHg.
Emphysema. Elastic fibers holding the small airways open are damaged, leading to difficulty breathing out. Additionally, the alveoli are damaged, so there is less surface area for gas exchange. This causes a decrease in PO2 in the blood.
Pulmonary edema. There is fluid in the alveoli, which increases the diffusion distance for gas exchange. This causes a decrease in PO2 in the blood.
Fibrotic lung disease. The alveoli are thickened, so there is a longer diffusion distance for gas exchange. This causes a decrease in PO2 in the blood.
Asthma. Increased resistance in the airways causes a decrease in airflow, which can lead to a decrease in PO2 in the alveoli and blood.
Ventilation-Perfusion Ratio
Ventilation (V) is the air reaching the alveoli per minute, and perfusion (Q) is the blood reaching the alveoli per minute. The V/Q ratio is around 0.8 in healthy individuals.
The body works to match ventilation and perfusion. When ventilation increases, perfusion also increases to maintain V/Q.
It also directs airflow and blood flow to optimize gas exchange. For example, the bottom of the lungs receives more blood due to gravity, so airways there dilate to increase ventilation.
Alveolar Gas Equations
The arterial partial pressure of oxygen and carbon dioxide can be calculated using the alveolar gas equations:
Where:
- is arterial partial pressure of CO2
- is alveolar partial pressure of CO2
- is barometric pressure
- is water vapor pressure
- is rate of CO2 production (mL/min)
- is alveolar ventilation (mL/min)
Note that we assume . This usually holds up unless diffusion between alveolar air and capillaries is limited.
Where:
- is arterial partial pressure of O2
- is alveolar partial pressure of O2
- is fraction of O2 in the alveoli
- is barometric pressure
- is water vapor pressure
- is rate of O2 production (mL/min)
- is alveolar ventilation (mL/min)
We expect to be around 40 mm Hg and to be around 100 mm Hg.
Oxygen Transport in Blood
In room air, oxygen and carbon dioxide act exactly the same. However, oxygen dissolves very poorly—it exerts 20x more partial pressure dissolved in water than CO2. This makes it difficult for blood to dissolve enough oxygen. Thus, most oxygen is actually bounded to hemoglobin.
The breakdown is:
- 1.5% dissolved in plasma
- 98.5% bound to hemoglobin
The oxygen-hemoglobin dissociation curve describes how the partial pressure of oxygen is related to the percent O2 saturation of hemoglobin. The curve is sigmoidal due to change in binding affinity and saturation. When one oxygen binds to hemoglobin, it increases the affinity of the remaining sites for oxygen.
When the blood leaves the lungs, the PO2 is around 100 mmHg and the hemoglobin is almost completely saturated. When the blood reaches the tissues, the PO2 level drops to around 40 mmHg, and the hemoglobin is around 75% saturated. This means that around 25% of the oxygen bound to hemoglobin is released to the tissues.
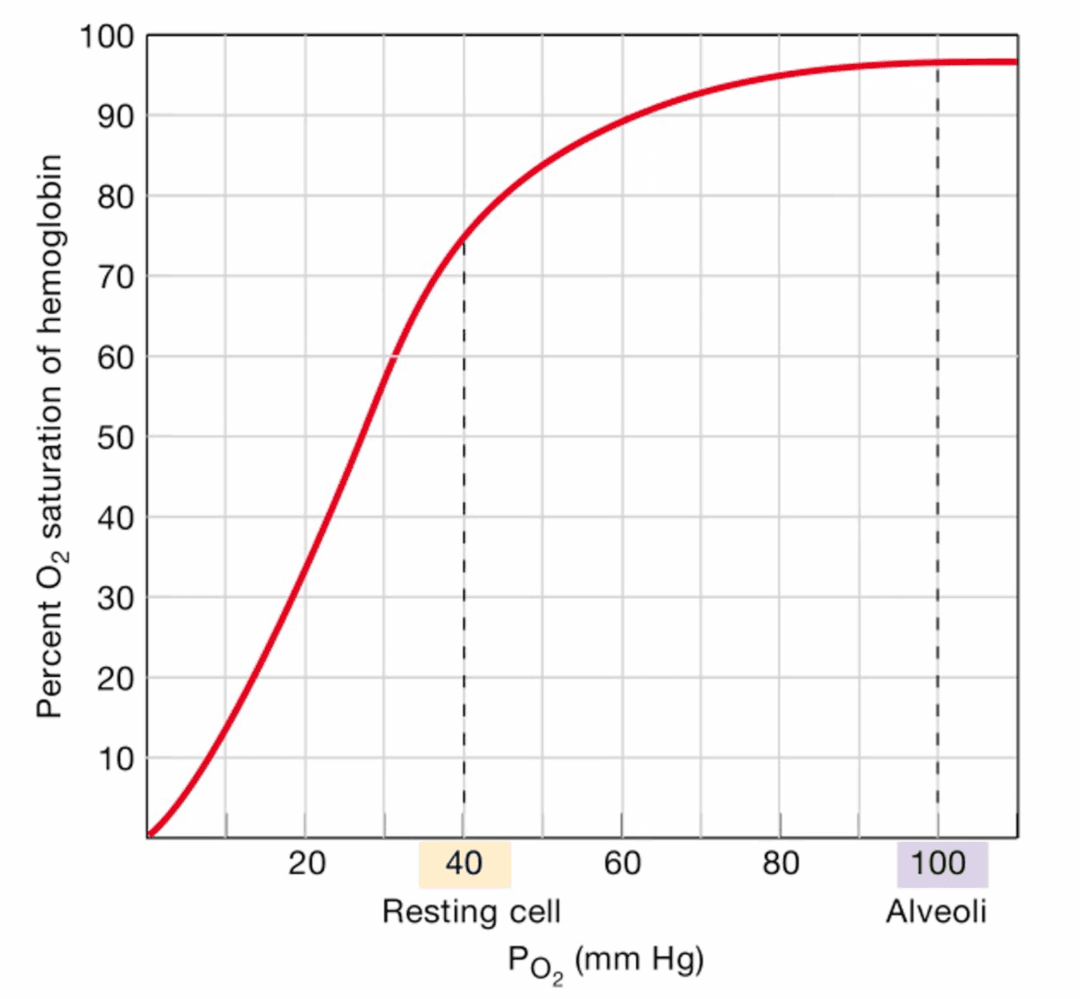
The Bohr effect states that the affinity of hemoglobin for oxygen decreases with
- Increased temperature
- Increased
- Increased [H+] (or decreased pH)
The Fick principle states that the rate of oxygen consumption by the tissues is equal to the product of cardiac output and the difference in oxygen content between arterial and venous blood:
Where:
- is the rate of oxygen consumption by the tissues (mL/min)
- is the cardiac output (mL blood/min)
- is the concentration of oxygen in arterial blood (mL O2/mL blood)
- is the concentration of oxygen in venous blood (mL O2/mL blood)
If the tissue is very metabolically active, then it will consume more PO2, leading to a lower point on the oxygen-hemoglobin dissociation curve. In addition, it will have a higher temperature and higher , leading to a Bohr shift to the right. Both of these effects will cause more oxygen to be released from hemoglobin, resulting in a lower concentration of . Thus, more oxygen will be delivered to the tissue. Note that changing is very difficult, but changing is much easier.
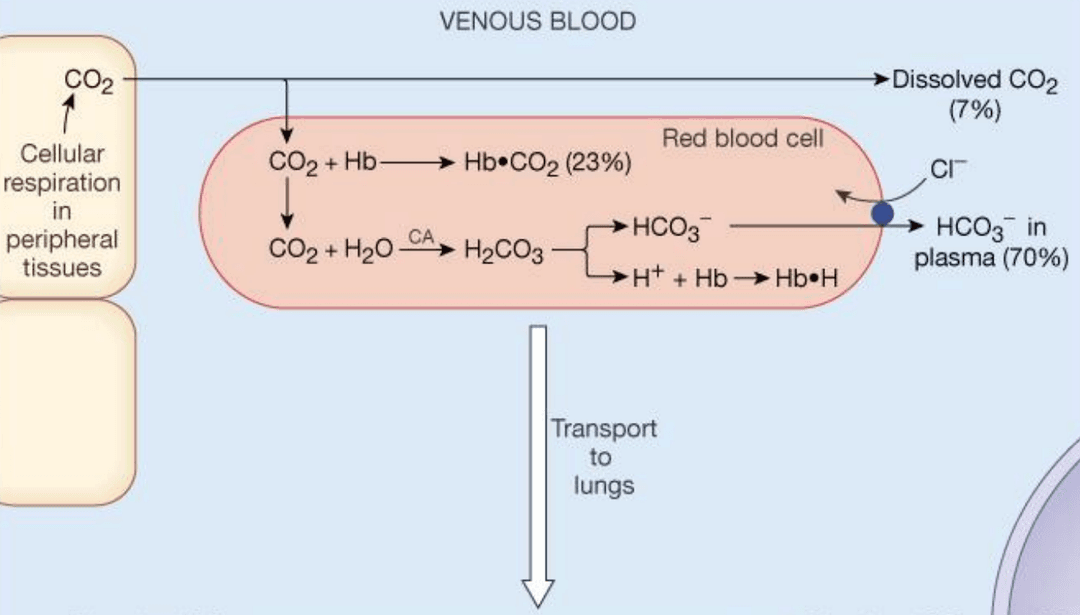
Carbon Dioxide Transport in Blood
Carbon dioxide is transported in the blood in three forms:
- 5% dissolved in plasma
- 10% bound to hemoglobin
- 85% as bicarbonate (HCO3-)
CO2 is converted to bicarbonate by the enzyme carbonic anhydrase in red blood cells. The reaction is:
By Le Chatelier's principle, when more CO2 is produced by the tissues, the reaction shifts to the right, producing more H+ and HCO3-. The H+ binds to hemoglobin, buffering the acidity. This also decreases hemoglobin's affinity for oxygen, causing more oxygen to be released to the tissues.
Control of Breathing
The brain monitors PO2, PCO2, and pH through chemoreceptors in the carotid bodies and aortic bodies. When PO2 decreases, PCO2 increases, or pH decreases, the brain increases ventilation to restore normal levels.
Rising CO2/falling pH is the first to regulate the 'urge to breathe'.
Renal System
Terms
Around 500 ml of plasma flows through the renal system per minute (RPF). Around 150 ml of that actually enters the glomerulus (GFR). As it travels through the renal system, around 124 ml of that is reabsorbed; only 1 ml is actually excreted as urine.
When the efferent arteriole is constricted, less fluid can move through the system, decreasing RBF. However, the constriction also increases hydrostatic pressure in the glomerulus, leading to more fluid being pushed into Bowman's space, increasing GFR.
| Change | RBF | GFR | FF |
|---|---|---|---|
| Afferent dilation | Increase | Increase | No change |
| Afferent constriction | Decrease | Decrease | No change |
| Efferent dilation | Increase | Decrease | Decrease |
| Efferent constriction | Decrease | Increase | Increase |
The filtration fraction is equal to GFR divided by RPF, and it tends to be around 0.3.
Clearance
The clearance of a substance is the volume of blood that is completely cleared of a specific substance by the kidneys per unit of time.
Where:
- is clearance
- is plasma concentration of the substance
- is urine concentration of the substance
- is urine flow rate
If the clearance of a substance is less than GFR, there is net reabsorption of that substance along the nephron. If the clearance of a substance is greater than the GFR then there is net secretion along the nephron.